How To Draw Lewis Structure For Ch2cl2
CH2Cl2 lewis structure, molecular geometry, polarity | Dichloromethane
Methylene chloride, also known as Dichloromethane (DCM), is an organic chemical compound. CH2Cl2 is the chemical formula for DCM. It is a colorless and volatile liquid with a sweetness smell. The chemical compound is naturally derived from the volcanoes, wetlands and other oceanic sources. It has many uses, but majorly it is used in the food industry. In this article, we volition know the construction, molecular geometry, applications and other chemical properties in detail.
CH2Cl2 Lewis structure
For agreement the properties and structure of any chemical compounds, including organic ones, its lewis structure is of the utmost importance. Lewis construction is a theory that helps in agreement the structure of a given compound, based on the octet rule. Co-ordinate to the octet rule, a molecule should have viii electrons in its outer crush to get inert or stable. For this chemical compound, there is 1 molecule of Carbon, two molecules of Hydrogen and 2 molecules of Chlorine.
To know the lewis structure, information technology is vital to discover the number of valence electrons in the chemical compound. Valence electrons are the sum total of the electrons every molecule has in their outer shell in a compound. These electrons include the ones that participate in bail formation as well as the ones that don't participate in forming bonds. The electrons that are involved in bond germination are chosen bonding pairs of electrons. Whereas the ones that practise not participate in forming whatever bonds are called alone pairs of electrons or non-bonding pairs of electrons.
In Lewis structure the lines correspond the bonds and dots represent the valence electrons. When nosotros talk about CH2Cl2, Carbon is less electronegative than Chlorine atoms. To empathize the Lewis structure lets first calculate the full number of valence electrons for Dichloromethane.
Carbon has four valence electrons, Hydrogen has i valence electrons and like all halogens, Chlorine has seven valence electrons.
Total number of Valence electrons = 4 + 2*i + 2*7
= 4+2+14
= twenty
There are twenty valence electrons in the compound, and iv bonds are formed. Key carbon atom forms two bonds with both Hydrogen and Chlorine atoms. Thus four valence electrons of Carbon, two electrons of Hydrogen and Chlorine each participate in the bond formation.
Hybridization of Dichloromethane

When two or molecules participate in the bond formation, their orbitals overlap due to the sharing of electrons. These overlapped orbitals are called hybrid orbitals. The bonds formed in Dichloromethane are covalent bonds. Central Carbon is hybridized as the molecule forms all the four bonds in the compound. An electron from the 22 orbital and three other electrons from 2p orbitals participate in forming bonds. Thus the hybridization of Carbon atom in CH2Cl2 is sp3.
Molecular Geometry of Dichloromethane
It is insufficiently easy to understand the molecular geometry of a compound after knowing its Lewis construction and hybridization. The arrangement of the molecules in this chemical compound is such that the Carbon atom is in the fundamental cantlet, 1 Hydrogen atom is on the upper topmost position and the other one is on the left side of the primal atom. Similarly, one chlorine atom is to the right of Carbon and the other 1 is i the downwardly position of the fundamental atom. As the hybridization is sp3, the molecular geometry of Dichloromethane becomes tetrahedral. The shape of the compound is a trigonal pyramidal.
Polarity of Dichloromethane
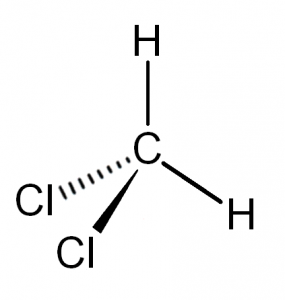
The polarity of any compound depends on the lone pairs of electrons and symmetry of the compound. It also depends on the electronegativity of the molecules participating in the formation of the compound. Here Hydrogen atom is less electronegative than chlorine cantlet and hence, there is a net dipole moment in the compound. Also, the arrangement of the bonded pairs is asymmetric, which makes Dichloromethane polar.
Physical Backdrop
Now that nosotros know all about the chemic properties and structures of CH2Cl2 let'southward have a look at its physical properties.
| Holding of the compound | Experimental values |
| Density of DCM | one.3226 g/cm iii |
| Molecular weight of DCM | 84.93 g/mol |
| Humid bespeak of DCM | 39.6 0 C |
| Melting point of DCM | -97.6 0 C |
Uses of Dichloromethane
- DCM is used equally a solvent in the nutrient industry and as a paint remover.
- It is also used as a degreasing agent.
- The compound is likewise used in the production of aerosol formulations.
Hazards of using Dichloromethane
- As the compound is highly volatile in nature, it can cause astute inhalation hazards. Prolonged exposure to DCM tin cause dizziness, fatigue, headache and much more as a result of astute absorption of the gas.
- DCM is metabolized as Carbon monoxide in the body that can atomic number 82 to Carbon Monoxide Poisoning in the torso.
- It has also been linked to diverse types of cancer and thus is a carcinogenic compound.
- The compound is also not safe for people with heart-related issues as it tin crusade aberrant heart rhythms and middle attacks when inhaled for an extended menstruum.
- In some cases, it can also irritate the olfactory organ and throat.
Last Remarks
I hope this article gives y'all detailed information about Dichloromethane. The compound has 20 valence electrons, out of which eight electrons participate in bond formation. It has sp3 hybridization and polar. DCM has tetrahedral molecular geometry and information technology is trigonal pyramidal in shape.
Source: https://geometryofmolecules.com/ch2cl2-lewis-structure-molecular-geometry-polarity/
Posted by: mcdonnellturper.blogspot.com


0 Response to "How To Draw Lewis Structure For Ch2cl2"
Post a Comment